Where does Türkiye stand in medical device manufacturing?
Diler Aslan
Based on my observations, knowledge, and experience spanning nearly fifty years, I believe we will not be able to advance to the next stage in medical device manufacturing.
Last week, I attended the Artificial Intelligence Symposium in Basic Oncology. Researchers presented their decision-making algorithms, machine learning, and artificial intelligence models. My takeaway from this symposium is that despite having very valuable human resources, the necessary ecosystem for product development has not yet been established in Türkiye.
The ecosystem is related to the product life cycle. Figure 1 shows the medical device life cycle as a product.
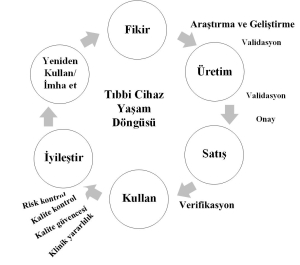
Figure 1. Medical device life cycle.
Figure 2 also shows the actors of the health ecosystem as an ecosystem.
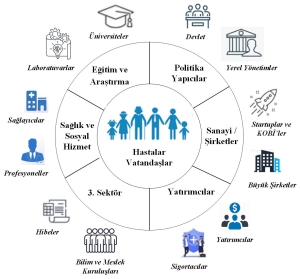
Figure 2. Healthcare ecosystem.
However, national progress can only be achieved if the role of each ecosystem actor in the product life cycle is determined and balanced. National strategies set the direction. Ecosystem actors, knowing their place, conduct their work through strategies, policies, projects, and processes they have defined.
Finding a coherent national health strategy document for Türkiye is quite challenging. Ministries select the strategies, policies, and measures they are responsible for from among the top-level policy documents. In accordance with Law No. 5018, they prepare institutional strategy plans for their own institutions. The implementation of these plans as strategic management is not clearly understood.
More importantly, corporate and national plans seem intertwined. It is very challenging for ecosystem actors to derive their own insights from these.
There are three separate plans regarding health: the strategic plans of the Turkish Ministry of Health, the Turkish Medicines and Medical Devices Agency (TİTCK), which is an institution of the Ministry of Health, and the Turkish Health Institutes Presidency (TÜSEB), which is also affiliated with the Ministry of Health.
There seems to be some confusion. Priorities are unclear. For example, there's a "National Artificial Intelligence Strategy," but a "National Data Strategy" is nowhere to be seen. Considering that data is used in the production of artificial intelligence, the most common saying comes to mind: "If garbage goes in, garbage comes out." This applies to all software within the scope of health information technologies, including artificial intelligence.
In human health and patient management, software used in decision-making is referred to worldwide as "Software as Medical Devices". in vitro Could they be classified as diagnostic (in-vitro invasive) medical devices? This needs clarification.
When examining the product lifecycle stages, it is observed that the most common stage in our country is the initial phase, which is the idea, research, and development phase… It appears from the presentations that the more advanced stages are not widely known.
The product manufacturing and launch phase is quite complex and comprehensive. Overcoming uncertainties in the resources, capabilities, and regulations of this cycle, especially human resources, is crucial for all stages.
These requirements are of great importance for medical devices, in vitro medical devices, and software used as medical devices.
It is essential for human health and patient safety.
Kind regards…
Prof. Dr. Diler Aslan
Pamukkale University Faculty of Medicine, Medical Biochemistry (2019-2019)
D-Tek Technology Development, Production, Consulting and Training Ltd. (2013-…)
October 3, 2025

Comments (0)